When patients ask me about “gummy bear implants,” they are usually asking two questions at once. First, they want to know what makes these implants different from older silicone implants. Second, they want to know whether “gummy bear” means better, safer, or more natural-looking. Those are fair questions, and they deserve a more precise answer than the nickname itself suggests.
The term “gummy bear implant” is really shorthand for a highly cohesive, form-stable silicone gel implant. The nickname came from the idea that the gel is thicker and holds its shape more like a gummy candy than a runnier silicone gel. The American Society of Plastic Surgeons describes gummy bear implants as form-stable implants that maintain their shape even if the shell is broken, and notes that the gel inside is thicker and the implants are firmer than traditional silicone gel implants.
That basic concept is what made these implants so exciting when they first became available in the United States. At the time, the attraction was obvious: if the implant could better hold its shape and was less likely to ooze even in the event of shell failure, that sounded like a meaningful technological step forward.
What I think is most important today, though, is not the nickname. It is understanding what the implant is actually designed to do, what tradeoffs come with that design, and where it fits into a modern breast augmentation conversation. “Gummy bear” is not a magic category. It is one implant characteristic among many that need to be weighed against anatomy, goals, feel, incision length, shape preference, and long-term considerations.
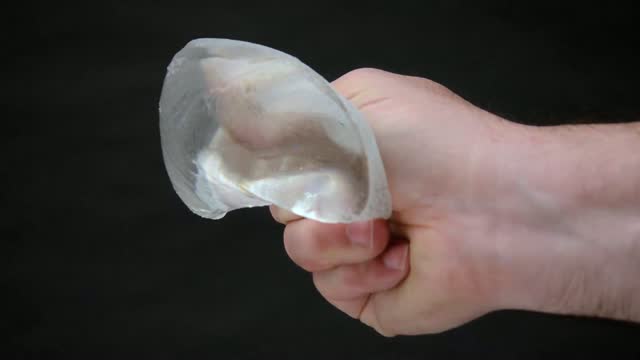
The new “Form Stable” implants from Sientra can actually be cut in half and squeezed, but still recover their shape, so oozing or leaking is very unlikely
Why Gummy Bear Implants Drew So Much Attention
The enthusiasm around gummy bear implants made sense. Highly cohesive silicone gel was presented as a more stable material, and in many ways, that was true. The FDA’s Summary of Safety and Effectiveness Data for Sientra’s silicone gel implants shows that the implants were supported by a large multicenter clinical study and approved by the FDA on March 9, 2012. That approval helped formalize the idea that more cohesive silicone gel was moving into mainstream U.S. practice.
The Appeal Was Shape Stability
The biggest selling point was shape stability. These implants were designed to better maintain their form, which appealed both to surgeons and to patients who wanted a device that behaved in a more predictable way inside the pocket. That stability also fed the common reassurance that if the shell were compromised, the gel would not simply behave like a liquid. ASPS still explains gummy bear implants in exactly those terms.
They Promised a More Advanced Silicone Option
Another reason they gained attention was timing. Silicone implants had returned to the U.S. market in 2006 after the FDA lifted the earlier restriction, and patients were already very interested in silicone again. The arrival of a more cohesive, form-stable version felt like the next evolution in that story rather than a completely separate invention.
They Fit a Very Real Patient Desire
Patients have long wanted implants that feel natural, look elegant, and reduce anxiety about rupture. Gummy bear implants spoke directly to that wish. That is part of why the term caught on so quickly, even though it is more of a consumer-friendly nickname than a precise medical category.
What “Gummy Bear” Actually Means in Practice
When I discuss this with patients, I try to strip away the marketing language and bring it back to fundamentals.
The Gel Is More Cohesive, and the Implant Is Firmer
Gummy bear implants use a thicker, more cohesive silicone gel than traditional silicone gel implants. That gives them more structural integrity, but it also means they are typically firmer. ASPS notes both of those points directly: they hold shape better and feel firmer than traditional silicone gel implants.
Not Every Cohesive Implant Is the Same
This is where the conversation gets more nuanced. Patients sometimes assume “gummy bear” refers to one exact implant made by one manufacturer. That is not really how the term is used now. Sientra currently describes its implant line as being built on cohesive silicone gel technology with multiple projection levels, gel types, and surface options, which reflects how broad the category has become.
The Nickname Can Oversimplify an Important Choice
I do not want patients choosing implants based on a catchy label alone. The real questions are: how soft or firm do you want the result to feel, how much upper-pole fullness do you want, how much structure do you need from the implant, and what implant characteristics best fit your tissues? “Gummy bear” may be part of that answer, but it is not the whole answer.
How They Compare With Traditional Silicone Implants
Patients often want a simple comparison, so I usually frame it this way: traditional silicone implants and highly cohesive silicone implants are both silicone implants, but they do not behave exactly the same.
Traditional Silicone Often Feels Softer
Traditional silicone gel implants tend to feel softer, and for many patients that softness is appealing. If the goal is a very soft, mobile feel and the anatomy is favorable, a less cohesive implant may still make perfect sense. FDA and ASPS materials both distinguish general silicone gel implants from more form-stable gummy bear implants, rather than treating them as interchangeable.
Gummy Bear Implants Prioritize Shape Retention
Highly cohesive implants are usually chosen when shape retention is a priority. That may be especially attractive in certain reconstructive or anatomically specific situations, or in patients who prefer a more structured result. But more structure is not automatically better. It is just different, and the value of that difference depends on the patient in front of me.
The Best Implant Is the One That Fits the Patient
This is why I do not treat implant selection as a popularity contest. A device can be technically advanced and still not be the best fit for a specific body. Breast augmentation works best when the implant matches the chest width, tissue thickness, skin envelope, and aesthetic goal—not when the patient simply picks the implant with the most exciting nickname.
The Shape Question: Round vs. Shaped Gummy Bear Implants
One source of confusion is that some people use “gummy bear implant” to mean shaped implant, while others use it more broadly to mean highly cohesive implant.
Shaped Implants Have a Distinct Design Logic
ASPS explains that shaped gummy bear implants have more projection at the bottom and taper toward the top. That design aims to mimic a more anatomic breast contour. In the right patient, that can be useful.
Rotation Is a Real Consideration
The tradeoff is that a shaped implant is not rotationally neutral. ASPS notes that if a shaped implant rotates, it can create an unusual breast appearance that may require another procedure to correct. That is one reason round implants continue to appeal to many surgeons and patients: they are more forgiving in that respect.
Round Cohesive Implants Often Make More Sense Aesthetically
This is one reason modern augmentation planning often favors round silicone implants, including cohesive ones, rather than assuming shaped implants are automatically more natural. ASPS notes that round implants can make breasts appear fuller and avoid the same concern about rotation because they are the same shape all over. In practical terms, that can be a very strong advantage.
The Incision and Technical Tradeoffs Patients Should Know
No implant technology comes without tradeoffs.
Gummy Bear Implants Usually Need a Slightly Longer Incision
ASPS states that placement of gummy bear implants requires a slightly longer incision in the skin. Patients do not always expect that, because they focus on the implant and forget that getting a firmer, form-stable implant into the pocket is also a technical issue.
Firmer Is Not Always Better
Patients sometimes hear “more advanced” and assume that means universally superior. I do not think that is the right way to think about it. A firmer implant can be an asset in some situations and a drawback in others. Feel matters. So does how the implant interacts with the patient’s own tissues. That is why I still think implant selection is highly individualized.
Implant Choice Is Still About the Whole Breast, Not the Device Alone
An implant does not exist in isolation. Pocket position, skin quality, natural breast tissue, chest shape, and whether the patient also needs a lift all affect the outcome. My augmentation approach has always reflected that principle: the implant is part of the plan, not the plan itself.
The Safety Conversation Has Become More Nuanced
I do not think it is responsible to discuss gummy bear implants today without acknowledging that implant safety conversations have become broader and more sophisticated.
FDA Counseling Requirements Matter
The FDA strengthened breast implant safety requirements in 2021, including stronger risk communication and patient decision materials. That matters because modern implant consent is more detailed than it used to be, and it should be. Patients deserve a fuller discussion of rupture, future surgery, surveillance, and implant-associated risks before proceeding.
Texture Is a Separate Conversation From Cohesiveness
Patients also need to know that cohesiveness and texture are not the same thing. The FDA notes that approved breast implants vary by fill, size, shell thickness, surface texture, and shape. That distinction matters because texture has had its own safety conversation, particularly around certain textured implants and BIA-ALCL.
The 2019 Allergan BIOCELL Recall Changed How Many Patients Think About Shaped Implants
The FDA requested a recall of Allergan BIOCELL textured breast implants and tissue expanders in 2019 because of the risk of BIA-ALCL, and it also stated that asymptomatic patients were not advised to remove or replace them routinely. This does not mean all cohesive implants are the same thing as recalled implants, but it does mean patients today are asking better questions about surface characteristics than they did when “gummy bear implants” first sounded like a simple technological upgrade.
Where Gummy Bear Implants Still Fit in My Practice
I still think highly cohesive implants have a real place. I also think they need to be chosen thoughtfully.
They Can Be a Good Option for the Right Anatomy
If I want more shape retention, more structure, or a more stable gel behavior, a cohesive implant may be very appealing. That is especially true when the patient understands what that means in terms of feel and incision tradeoffs.
They Are Not Automatically My First Choice for Everyone
At the same time, I do not think every augmentation patient needs or wants the firmer feel associated with more form-stable implants. Some women want softness above all else. Others want a rounder, fuller look that may be served beautifully by a different silicone implant. Good augmentation planning is always more individualized than the nickname suggests.
Sometimes the Better Conversation Is About the Entire Augmentation Plan
Patients often come in asking specifically for a certain implant, when what they really need is a better understanding of the whole augmentation plan. Implant type, incision, pocket placement, whether a lift is needed, and what kind of result they actually want all matter more than simply asking for “gummy bears.” That broader discussion is still the one I find most useful.
My Bottom Line on Gummy Bear Implants
Gummy bear implants are real, useful, and in many cases very good implants. The term refers to highly cohesive, form-stable silicone implants that better hold their shape and are generally firmer than traditional silicone gel implants. That is the real technological story behind the nickname.
But I would not want a patient to hear the nickname and assume it answers the whole implant question. It does not. The meaningful questions are still the same ones they have always been: how do you want your breasts to look and feel, what does your anatomy require, what tradeoffs are you comfortable making, and which implant characteristics best serve that goal? The best augmentation result comes from matching the implant to the patient—not from chasing the most memorable label.
Curious About Gummy Bear Implants? Let’s Decide Whether They’re Actually Right for You
If you are interested in breast augmentation and wondering whether gummy bear implants make sense for your body and your goals, the best next step is a thoughtful consultation. I can evaluate your anatomy, explain how cohesive implants compare with other silicone options, and help you decide whether the advantages of a more form-stable implant are truly the right fit for the result you want.





Leave a Reply